
| RELATED INFO |
| * Ji-Xin Cheng |
| * Weldon School of Biomedical Engineering |

April 14, 2009
Findings show insulin - not genes - linked to obesity
WEST LAFAYETTE, Ind. -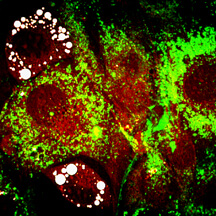 |
Learning the precise mechanism responsible for fat storage in cells could lead to methods for controlling obesity.
"Insights from our study also will be important for understanding the precise roles of insulin in obesity or Type II diabetes, and to the design of effective intervention strategies," said Ji-Xin Cheng, an assistant professor in Purdue University's Weldon School of Biomedical Engineering and Department of Chemistry.
Findings indicate that the faster a cell processes insulin, the more fat it stores.
Other researchers have suggested that certain "fat genes" might be associated with excessive fat storage in cells. However, the Purdue researchers confirmed that these fat genes were expressed, or activated, in all of the cells, yet those cells varied drastically - from nearly zero in some cases to pervasive in others - in how much fat they stored.
The researchers examined a biological process called adipogenesis, using cultures of a cell line called 3T3-L1, which is often used to study fat cells. In adipogenesis, these cells turn into fat.
"This work supports an emerging viewpoint that not all biological information in cells is encoded in the genetic blueprint," said Thuc T. Le, a National Institutes of Health postdoctoral fellow at Purdue who is working with Cheng. "We found that the variability in fat storage is dependent on how 3T3-L1 cells process insulin, a hormone secreted by the pancreas after meals to trigger the uptake of glucose from the blood into the liver, muscle or fat cells."
The findings are detailed in a research paper appearing online in the journal PLoS ONE, published by the Public Library of Science, a non-profit organization of scientists and physicians.
"This varied capability to store fat among genetically identical cells is a well-observed but poorly understood phenomenon," Cheng said
The researchers determined that these differences in fat storage depend not on fat-gene expression but on variations in a cascade of events within an "insulin-signaling pathway." The pathway enables cells to take up glucose from the blood.
"Only one small variation at the beginning of the cascade can lead to a drastic variation in fat storage at the end of the cascade," Cheng said.
The researchers conducted "single cell profiling" using a combination of imaging techniques to precisely compare fat storage in cloned cells having the same fat genes expressed.
Single cell profiling allows researchers to precisely compare the inner workings of individual cells, whereas the conventional analytical approach in biochemistry measures entire populations of cells and then provides data representing an average.
"In this case, we don't want an average. We need to find out what causes fat storage at the single-cell level so that we can compare one cell to another, " Le said. "By profiling multiple events in single cells, we found that variability in fat storage is due to varied rates of insulin processing among cells."
The cell culture used in the research contains cloned mice fibroblast cells.
"This particular type of cell culture has been used to study the molecular control of obesity for the past 35 years," Cheng said. "Researchers have observed tremendous variability in how much fat is stored in cells with identical genes, but no one really knows why. Our findings have shed some light on this phenomenon."
The researchers used a specialized imaging method called coherent anti-Stokes Raman scattering, or CARS, combined with other techniques, including flow cytometry and fluorescence microscopy.
"This multimodal imaging system allows us to correlate different events, like fat storage, gene expression and insulin signaling," Le said. "We can monitor these different events at the same time, and that's why we can determine the mechanism at the single-cell level."
Insulin attaches to binding sites on cell membranes, signaling the cells to take up glucose from the blood. Cells that are said to be resistant to insulin fail to take up glucose, the primary cause of Type II diabetes, a medical condition affecting nearly 24 million Americans. About two-thirds of U.S. adults are overweight, and nearly one-third obese.
The research, which has been funded by the National Institutes of Health, is ongoing. Future work may seek to pinpoint specific events in the insulin-signaling cascade that are responsible for fat storage.
Writer: Emil Venere, (765) 494-4709, venere@purdue.edu
Sources: Ji-Xin Cheng, (765) 494-4335, jcheng@purdue.edu
Thuc T. Le, (765) 496-9717, let@purdue.edu
Purdue News Service: (765) 494-2096; purduenews@purdue.edu
Note to Journalists: An electronic copy of the research paper is available from Emil Venere, (765) 494-4709, venere@purdue.edu
PHOTO CAPTION:
Purdue researchers have uncovered new evidence that factors other than genes could cause obesity, finding that genetically identical cells store widely differing amounts of fat depending on subtle variations in how cells process insulin. Here, insulin (green) is present in cells with no fat storage and absent in cells with fat storage at two days after insulin addition. This observation indicates faster insulin processing rates in cells with fat storage. Fluorophore-labeled insulin (green) is visualized with fluorescence imaging, and fat is visualized with coherent anti-Stokes Raman scattering - or CARS - imaging (red/white). (Weldon School of Biomedical Engineering, Purdue University)
A publication-quality photo is available at https://www.purdue.edu/uns/images/+2009/cheng-fatgenes.jpg
Single-Cell Profiling Reveals the Origin of
Phenotypic Variability in Adipogenesis
Thuc T. Le1 and Ji-Xin Cheng 1, 2
1Weldon School of Biomedical Engineering;
2Department of Chemistry
Corresponding author: Ji-Xin Cheng, Weldon School of Biomedical Engineering, Purdue University, West Lafayette, IN 47907, Tel 765 494 4335, Fax 765 494 1193,
Email: jcheng@purdue.edu
Phenotypic heterogeneity in a clonal cell population is a well-observed but poorly understood phenomenon. Here, a single-cell approach is employed to investigate non-mutative causes of phenotypic heterogeneity during the differentiation of 3T3-L1 cells into fat cells. Using coherent anti-Stokes Raman scattering microscopy and flow cytometry, adipogenic gene expression, insulin signaling, and glucose import are visualized simultaneously with lipid droplet accumulation in single cells. Expression of adipogenic genes PPARγ, C/EBPα, aP2, LP2 suggests a commitment to fat cell differentiation in all cells. However, the lack of lipid droplet in many differentiating cells suggests adipogenic gene expression is insufficient for lipid droplet formation. Instead, cell-to-cell variability in lipid droplet formation is dependent on the cascade responses of an insulin signaling pathway, which includes insulin sensitivity, kinase activity, glucose import, expression of an insulin degradation enzyme, and insulin degradation rate. Increased and prolonged insulin stimulation promotes lipid droplet accumulation in all differentiating cells. Single-cell profiling reveals the kinetics of an insulin signaling cascade as the origin of phenotypic variability in drug-inducible adipogenesis.
To the News Service home page
